Fixing Broken Memory
Dr. Robert E. Hampson
© 2018 Lightspring, image licensed from Shutterstock
Memory loss: there can be no more tragic condition for humans, particularly in Alzheimer's Disease where memory loss is progressive and debilitating. AD patients initially show problems with “working memory” (new, temporary memories for a specific purpose, such as remembering where you parked your car), then more serious memory deficits set in resulting in loss of “reference memory” (established information such as personal history and facts learned in school). Eventually, the memory loss is so severe, the patient loses all sense of self, and becomes like a stranger to family members, having already forgotten the names and faces of their loved ones.
Loss of memory function can be temporary, such as amnesia, or progressive and debilitating, such as the example of dementia given above. It can accompany head injury or stroke, result from disorders such as Parkinson's Disease, Alzheimer's and epilepsy, or occur as a consequence (intended or not) of medication and drugs of abuse. Much research on human memory has focused on identifying brain areas that can produce amnesia and/or dementia when damaged. Therapeutic measures to counter memory loss have focused on protecting those brain areas, correcting metabolic or chemical imbalances, and in rare cases, testing drugs that improve the neurochemical processes underlying memory and promote enhanced memory abilities. Unfortunately, few of these research directions deal with the underlying issues of replacing or restoring the actual function of memory performed by assemblies of neurons (electrically active cells) in the brain.
Until now.
For the past 35 years, I have been privileged to be a member of a team that has examined basic functions of the brain underlying memory. This spring, we announced the successful testing of a system with the potential to become a prosthetic device for restoration of memory function in humans [http://iopscience.iop.org/article/10.1088/1741-2552/aaaed7]. The unique capability of this system is the potential for bypassing memory-associated brain areas that have been damaged and to take over information processing for the damaged areas. The work was supported in part by the Defense Advanced Research Projects Agency (DARPA) via their Restoring Active Memory (RAM) program directed by Dr. Justin Sanchez. The project is also the culmination of nearly twenty years of DARPA, NSF, and NIH-supported research in various animal models prior to extending the testing to humans.
To explain the magnitude of the problem of "Broken Memory" requires some background in what physicians and scientists know about memory function. Readers may also be interested in a Baen nonfiction essay "Remember to Remind Me" from 2015 [http://www.baen.com/remember]. Furthermore, to understand our team's prosthetic approach will require a diversion into some of the key researchers and their findings, as well as competing theories and proposed treatments. After the basics, we will discuss the timeline for getting from this “basic science” to the current studies. No announcement of proposed treatment would be complete without a discussion of alternatives—such as drugs, memory training, and even alternate prosthetic proposals. Finally, we will discuss the current state of the art in the context of advanced theories and treatments as put forward by science fiction writers and futurists.
Human Memory—and How to Damage It
Human memory is generally categorized in different ways in order to identify both the function and the involvement of different brain areas. The most common category is short-term vs. long-term memory. Short-term memory is information that only needs to be retained for a brief duration—such as the interval between reading and dialing a phone number. Such information is rapidly and easily forgotten. Long-term memory is just what it sounds like: information that is stored in our memory more-or-less permanently, such as the names of friends and family, or information we learned in school. Two alternative terms that mean close to the same thing as short- and long-term memory are “working memory” and “reference memory.” Working memory is a particular type of short-term memory in which the information is retained only briefly, used for a purpose and then discarded. Reference memory is facts and figures that we learn through repetition and long exposure—in other words, long-term memory such as the names of U.S. Presidents or states, how to do algebra, or read Latin. Short-term and working memory are often synonymous, but researchers quibble over the time lengths involved; my studies involve intervals of up to two minutes, yet others may concentrate on lengths of less than ten seconds. One area of agreement, however, is that short-term memory has the potential to become long-term memory through several processes that have actually been covered in the “Remember to Remind Me” essay mentioned and linked above. Memory of any duration can also be categorized as "Episodic" meaning that information that is experienced one or more times; or “Declarative,” which is essentially information of descriptive and mechanistic nature. Thus, Episodic memory is related to what we experience, while Declarative memory is related to what we learn. Note that none of these labels is entirely appropriate to skill memory such as riding a bike, driving a car, or speaking a language. Skill memory is a form of reference memory, but there are several important differences involving how we learn skills by repetition.
What then, is the importance of these different types of memory and the brain? Doctors and scientists have long known that certain types of head injury could damage some types of memory, but it was not until the case of “H.M.” in the 1950s that neuroscientists began to truly understand the particular role of the hippocampus in the various functions of memory. In 1957, Henry Molaison underwent surgery in hopes of stopping his epileptic seizures. The surgery would remove most of the medial temporal lobe—an area extending down and forward on the left and right sides of the brain. Make a fist with your hand, place your thumb along the outside of the first finger, don't curl it inward or across the other fingers, then hold the hand thumb down. That's a rough likeness of one-half of the brain, and your thumb represents the temporal lobe. On the inner side of the temporal lobe is a structure called the “hippocampus,” Latin for “seahorse” (due to the fact that early anatomical descriptions noted a similarity to the curve of a seahorse's tail). The operation successfully stopped the seizures, but it left the patient with a profound amnesia. Most importantly, the type of deficit was “anterograde amnesia,” a failure of short-term memory in which the patient is unable to retain temporary information for more than a few minutes, and in turn, unable to turn that information into permanent (reference) memories. The fact that H.M.'s amnesia was so similar to the diagnosis of progressive dementia described by Professor Alois Alzheimer in 1901, led scientists to closely examine the pathology of Alzheimer's Disease and carefully consider the role of the hippocampus in memory.
There are many brain areas involved in memory; injury to the parietal lobes (the back of the hand in our fist-like model of the brain) can impair recall and reference memory. Specific brain areas for language and speech combine sensory (sight and sound), motor (speech and writing) and memory functions (and are located roughly equivalent to the “knuckles” in our hand model). Specialized amnesias such as “aphasia” (forgetting words) or “alexia” (inability to read) can result from damage limited to those areas. However, the most profound association between memory and brain area resides in the role of the hippocampus to mediate the formation of new memory, making it extremely important for short-term, working memory, and/or episodic memory.
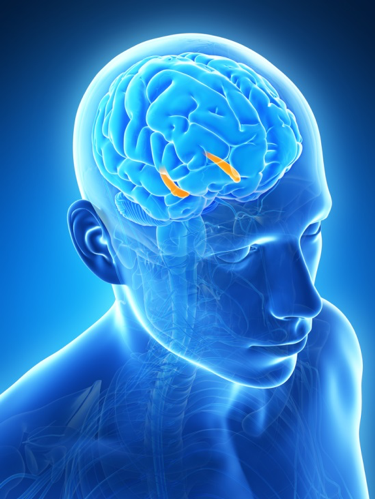
The hippocampus (orange) lies along the inner edge of the temporal lobe on both sides of the brain. © 2018 Sebastian Kaulitzki, image licensed from Shutterstock
Codes for Memory in the Brain
Once brain areas were identified with particular functions, neuroscientists started looking closer at how the individual neurons (brain cells) were involved. For the “sensory” and “motor” areas of the brain, this study had been well underway at the time when the hippocampus came to center stage in the 1960s. However, the techniques used to study the association between brain, senses (touch, taste, smell, hearing, vision) and muscle movement were not appropriate. Neurosurgeon Wilder Penfield mapped much of the brain's relationship with the body by using small electrical charges to activate parts of the brain and either observe what body part moved (motor) or asking the patient what they felt (sensory). However, that approach yielded very few results when applied to memory and the hippocampus.
Then in 1971, researchers John O'Keefe and Jonathan Dostrovsky noticed that certain cells in the hippocampus of rats were active only when the rat was in a particular position in the cage or test chamber [https://doi.org/10.1016/0006-8993(71)90358-1]. The finding led to nearly 50 years of research into "place cells" in the hippocampus, correlating their activity with various elements of location. Cells have been identified with preferences to corners, edges, head directions, body directions, both future and past movements. O'Keefe and colleagues later referred to this as a "cognitive map" of the environment. In 1994, Matt Wilson and Bruce McNaughton demonstrated that place cells were not only involved in a physical representation, but that they were an important component of memory [https://doi.org/10.1126/science.8036517]. Utilizing a track that limited a rat's movement through the environment to specific lanes, thus a specific sequence activating place cells, the team demonstrated that the same cells were re-activated during sleep in the same sequence as when the animal had passed through them in the test session. By making the rat's behavior and reward dependent on remembering a previous sequence of movement, and then manipulating whether this sleep-replay could occur, Wilson, McNaughton and their colleagues were eventually able to demonstrate that the replay was an important phase of memory formation.
My own research started out in a lab that studied the hippocampus, and for years, I, too, recorded place cells and mapped the position correlates of hippocampal neurons. However, we soon discovered that there was more than just spatial information represented by the hippocampus. By this point, place cells had been identified in mice, rats, guinea pigs, cats, dogs, monkeys and humans; yet simply mapping position did not mesh with the known involvement of the hippocampus in encoding all types of memory, as shown by the case of H.M.
From Rodents to Primates to Humans
In the late 1960s/early 1970s the neurophysiology laboratory of Dr. Gary S. Lynch at the University of California at Irvine was deeply involved in understanding the role of the hippocampus in memory. Many theories had been put forward: that the hippocampus acted as an amplifier, that it detected mis-matches between new and old information, that it fulfilled a “pattern completion” role (filling in the gaps in partial data), or even that it had a purely olfactory function (sense of smell). Lynch believed that the hippocampus was processing information related to memory, and one of the young researchers, Dr. Sam A. Deadwyler, started experiments designed to find out exactly how hippocampal neurons encoded information. Soon after, Dr. Theodore W. Berger at the University of Pittsburgh was studying how hippocampal neurons altered their function when information was repeated, strengthening some connections, and altering others to produce a new result when familiar inputs were received. Deadwyler would end up in the Department of Physiology and Pharmacology at Wake Forest School of Medicine, and Berger in the Department of Biomedical Engineering at the University of Southern California, but their eventual long-distance collaboration would bear significant fruit in the form of a model of hippocampal function in memory that shaped my own career, and underlies the recent memory prosthetic paper.
In 1996, I was part of Deadwyler's lab, and studied the activities patterns of collections of hippocampal neurons while rats performed a task requiring short-term memory. These “ensembles” or collections of neurons fired in different patterns in both time and “space” (in this case, space referred to different neurons distributed throughout the hippocampus) depending on the type of behavior required in the task. More importantly, we could identify patterns that clearly distinguished between the various stimuli in the task including position (similar to place cells), behavioral responses, and what part of the task was occurring. Analyses based on the neural ensemble activity alone without looking at those variables could identify and even “predict” some of those variables even before they occurred [http://www.jneurosci.org/content/jneuro/16/1/354.full.pdf]. In fact, the patterns could even identify when the rat was about to make an error in the task [https://www.ncbi.nlm.nih.gov/pmc/articles/PMC33635/]. Over the following ten years, Deadwyler's laboratory refined the analysis to identify the “codes” and improved the ability to predict correct and error responses. The studies moved from rats to monkeys, and would eventually move into human studies, but a few other factors had to come into play, first.
During this same time interval, Berger teamed up with neuroscientist Dr. Vasilis Z. Marmarelis, who had developed a mathematical approach to apply nonlinear systems analysis to neural function. Nonlinear systems are often mislabeled as “chaotic.” More accurately, they do not behave in a regular, exactly repeating manner; rather, they form patterns that appear to recur, but with significant differences each time (consider the annoying lack of pattern in a dripping faucet!). Berger and Marmarelis applied the nonlinear analyses to study the activity of slices of hippocampus kept alive in cell-culture conditions that allowed recording in isolation from all other brain activity [https://doi.org/10.1016/S0925-2312(98)00160-X]. Even in isolation, they noted that activity of the hippocampal neurons was highly dependent on temporal and spatial (in this case, spatial = distribution of neurons and connections across the hippocampal slice) activity of both the input signals and the activity of other neurons in the tissue slice.
Deadwyler and Berger compared notes at several scientific conferences throughout the 1990s and by 2002 had decided to team up to apply the nonlinear model to study the predictive patterns of hippocampal neurons with respect to memory. At the time, the Defense Advanced Research Projects Agency had announced a program to promote Brain Machine Interface research. Deadwyler and Berger's team joined with other researchers on the project and began to extend the modeling from Berger's hippocampal slices, to Deadwyler's behaving rat studies, and eventually into studying memory in other species. By 2005, Berger had published his concept for a prosthetic device in Toward Replacement Parts for the Brain (MIT Press, 2005). The device would record from the inputs to the hippocampus, compute the nonlinear prediction of hippocampus output, then stimulate "downstream" from the hippocampus with that prediction, effectively "bypassing" a damaged hippocampus to restore the function of encoding information for memory processing.
In 2011, the team published their first report that the prosthetic-style stimulation improved memory performance in rats with an intact (undamaged) hippocampus [https://doi.org/10.1088/1741-2560/8/4/046017]. But what about a damaged hippocampus? Would the method work where the hippocampal signals were confused or incomplete? In short, yes. Part of the test of the model was to use a drug to partially inactivate the hippocampus and see if the model stimulation would improve or restore the memory function . . . and it did!
With a successful demonstration in rats, the team focused on progressing to a system that could be applied to human memory. However, primate memory is more complex than in rodents, and more than one brain area is involved. In fact, it was mentioned above that some laboratories differ on the definition of “short-term memory” with some laboratories focusing on very short intervals of 1-10 seconds, with others focusing on a longer interval out to 120 seconds. Studies in nonhuman primates traditionally ascribe the very short duration memory to the prefrontal cortex—in the front part of the brain, and the longer duration memory to the hippocampus. By this point, I had taken a more active role in directing the study, so Deadwyler and I initially tested Berger's model in the prefrontal cortex of rhesus monkeys [https://doi.org/10.1088/1741-2560/9/5/056012] and then the hippocampus [https://doi.org/10.1088/1741-2560/10/6/066013] with success in both approaches. Along the way, Berger and his colleague Dr. Dong Song had adapted the original model from rodent hippocampus to primate prefrontal cortex and hippocampus, and then began to adapt the model for humans [https://doi.org/10.1109/TNSRE.2016.2604423]. The details were different, but the essential model remained a means of predicting hippocampal outputs from inputs to bypass a damaged hippocampus. To restore memory function even when memory was already in decline, the models were trained to discriminate between successful memory (correct trials) and unsuccessful memory (error trials) such that the residual memory function could be reinforced by the operation of the model.
It was now time to test the system in humans.
Nootropic Drugs, Biofeedback and Deep Brain Stimulation
There are many proposed ways to improve human memory, and even rescue it from decline. Science Fiction typically invokes a miracle drug that can “speed up the brain” or similarly “promote the use of more than 10 percent of your brain!" In fact, much of this is nonsense; it is neither the speed of processing, nor the amount of brain in use that determines memory accuracy and efficiency. In fact, humans use nearly all of their brain, all of the time. Inactive tissue is, frankly, dead. The brain remains active all the time, just not particularly involved in all functions at any given time. Just because a person is remembering a song, doesn't mean their vision and touch processing areas aren't active. The brain processes a lot of information, all the time. As you read this essay, you may not be thinking about the sensation of clothes on your skin, seat of the chair, position of your legs, but the sensation is there if you choose to pay attention to it. Likewise, problems with memory fall into two categories: attention to details, and disease/injury.
“Brain training” exercises can be found on the internet and even as smartphone apps. The hype promises an increase in mental acuity and a defense against senility and dementia. Memory tricks and training encourage the user to learn to pay attention to details and make information more memorable. Biofeedback works the same way, it encourages one to adopt brain states that are conducive to attention to detail, and hence improve memory. This does help, and people can be trained to remember with greater success, but the technique has no benefit when the hippocampus and other brain areas are damaged by head injury, stroke, epilepsy, Parkinson's Disease or Alzheimer's disease.
There are pharmacological means of making the biochemical (and electrical) interactions of neurons more efficient. “Nootropic” drugs enhance the communication within and between neurons, focusing attention and increasing the apparent “power” of memory and cognition. The recent movie Limitless is actually based on studies of a drug called “aniracetam,” which makes neurotransmission—the chemical communication between neurons—more efficient. At the same time, movies that invoke the “you only use 10 percent of your brain” concept or postulate a dramatic instantaneous “evolution” to a higher state of being (such as Lucy) are pure fantasy. Thus, there are nootropic drugs that can have a benefit in patients with memory disorders. Memantine is one of those drugs, and it is currently prescribed to Alzheimer's patients to slow down the memory loss associated with the disease. Unfortunately, we still don't have a way to totally arrest the decline and save a patient's memory.
Another popular science fiction trope is electrical stimulation of the brain. Many people probably equate brain stimulation with electro-convulsive shock therapy (ECS). The image of mental patients being shocked off of a table is a visceral one, and hard to erase. The truth is that milliampere and microampere stimulation is a very valuable tool in treating brain disorders, unlike the tens of amperes and volts associated with ECS. Cochlear implant devices have restored the sense of hearing to patients for over thirty-five years. Retinal implants have been FDA approved for five years, and promise to do the same for patients with loss of vision. Transcranial magnetic stimulation and transcranial direct current stimulation apply magnetic fields or electrical current (which generates magnetic fields) to the scalp to influence neural activity. While both techniques produce changes in brain activity and behavior, the exact mechanisms of action are largely unknown, and more research is needed. Deep brain stimulation (DBS), uses a milliampere rhythmic electrical stimulation to the thalamus (a structure responsible for relaying sensory and motor information between the body and the brain) to alleviates many symptoms of Parkinson's disease. DBS is considered the “pacemaker of the brain,” and dates back to 1987, with the procedure becoming much more common in the late 1990s and early 2000s. Many hospitals and medical centers have each implanted thousands of patients with DBS electrodes and stimulators over the past twenty years!

Jan Scheuermann feeds herself a chocolate bar, using a robotic arm signals from her brain [Photo by University of Pittsburgh, used with permission.]
Neuroelectrical prosthetics are becoming much more common, and a science fiction author of my acquaintance likes to “complain” that scientists and doctors are making it too hard to write SF, since we keep making the wildest predictions of SF come true! Over the past ten years we have seen retinal prosthetics, brain-controlled robotic arms (above), and even fuzzy cat ears that wiggle in response to brain waves. But what about memory? Can DBS or a technique similar to it restore or even facilitate memory?
Brain Stimulation and Human Memory
It is an acknowledged fact that sometimes the most astounding findings in science occur by accident. With thousands of patients receiving DBS implants, sometimes the electrodes are not precisely in the desired position. However, as a consequence of these sub-optimal placements, clinicians and researchers have learned that DBS stimulation can have effects on other brain systems, potentially relieving anxiety and depression, calming Obsessive Compulsive Disorder, and even assist in treating eating disorders. An electrode placement in one patient was particularly surprising. In the course of studying a patient with electrode placement close to the fornix (one of the brain structures containing major inputs to the hippocampus), as well as subsequent patients with deliberate placement of DBS stimulating electrodes in the fornix, Toronto Neurosurgeons Adrian Laxton, M.D. and Andres Lozano M.D., Ph.D. determined that DBS-style stimulation slowed the progression of Alzheimer's Disease [https://doi.org/10.1016/j.wneu.2012.06.028]. In California, UCLA psychologist Nanthia Suthana, Ph.D. and neurosurgeon Itzhak Fried, M.D., Ph.D. stimulated entorhinal cortex (another structure with strong inputs to hippocampus) with DBS-like signals and also enhanced learning and memory [https://doi.org/10.1016/j.neuroimage.2013.07.066].
The Defense Advanced Research Projects Agency was already involved in the advancement of brain-controlled prosthetics. Program manager Colonel Geoffrey Ling, M.D. (U.S. Army, retired) launched the Revolutionizing Prosthetics program which culminated in the brain controlled robotic arm shown above. The program went on to develop wearable artificial limbs that begin to fit the definition of “bionic.” A term first coined by Jack Steele, M.D., meaning “lifelike,” it gained a foothold in science fiction, but has since spread into common use [see “On the Road to the Brainships: A Look at the Current Science of Interfacing the Brain,” http://www.baen.com/brainships]. Building on the results from several DARPA-funded research programs, including ours, plus the results by Laxton and Lozano, and Suthana and Fried, DARPA initiated the “Restoring Active Memory” (RAM) program in 2014 under the leadership of Justin Sanchez, Ph.D.
DARPA RAM has funded two different approaches to the problem of facilitating and restoring human memory. One is a very DBS-like approach led by Michael Kahana Ph.D. and Daniel Rizzuto Ph.D. at the University of Pennsylvania. The UPenn team includes neurosurgeons and researchers at nearly a dozen hospitals, research institutions and instrument companies. I lead the other group with laboratory and clinical colleagues at Wake Forest Baptist Medical Center and USC, using an approach based on the work pioneered by Berger and Deadwyler.
The UPenn team was actually first into press with their results in which they studied hundreds of patient brains to detect changes in the surface and subsurface brain activity associated with correct and incorrect memory functions. The recording approach is based on electroencephalograms (EEG) that incorporate the activity of large assemblies of neurons, and do not always correlate to single brain areas (not that brain always has distinct areas, either!). Spectrographic analysis of this data revealed high frequency activity in the temporal lobe when memory was functioning correctly, which was absent or weaker when memory errors were committed. These results suggested that it was possible to detect a “brain state” corresponding to strong vs. weak memory. Kahana and Rizzuto's colleagues designed a study to detect the “bad memory” state and to reinforce the “good memory” state via stimulation applied to the surface of the brain over the area where the changes in EEG signal had the greatest correlation with memory errors. The stimulation was applied during the time period when the hippocampus “encodes” information for later recall; the recall of that information proceeded naturally, with no stimulation, allowing the team to test how the “good memory” state influenced overall memory function. The approach was successful and the team demonstrated memory improvements of around 15 percent in memory function and recall [https://doi.org/10.1038/s41467-017-02753-0].
An interesting corollary finding by the UPenn team and other researchers was that stimulation applied directly to the hippocampus was unsuccessful. In fact, direct stimulation of the hippocampus with a DBS-like signal impaired memory, leading both teams to look at stimulation sites outside of hippocampus. This finding was of significance to the WFBMC/USC team, since our stimulation was applied directly to the hippocampus. However, our techniques were fundamentally different both in the theory and type of stimulation.
Berger's theory was based on the detection of discrete neuron firing in the hippocampus. Deadwyler's team had identified neurons which changed their electrical activity in response to the events of a behavioral trial—i.e. was it the encoding or the recall phase? Did the subject simply view the stimulus or did they have to make a response? Was the position to the left or right? Was there one picture or two or three? Each neuron studied responded to a particular feature or combination of features, but no one neuron was capable of responding to (and hence encoding) every feature of a trial or item to be remembered. Encoding of the whole information required multiple neurons—an ensemble in neuroscience terms—and the details of ensemble activity are lost when recorded as EEG-style signals far from the neurons that produce them. Furthermore, Berger had demonstrated that ensemble firing was a product of nonlinear dynamics within and between neurons. Thus, fixed frequency stimulation would disrupt that memory-dependent activity. What was needed instead was a pattern of stimulation that mimicked the nonlinear ensemble codes underlying memory function.
Our project recruited patients who were undergoing a surgical procedure to map the occurrence of epileptic seizures. These patients had electrodes implanted into hippocampus to determine where their seizures originated, with some occurring within hippocampus, and others originating in neighboring brain areas. The patients also had baseline memory that ranged from excellent to impaired, due to their disease. The WFBMC team was able to record neuron ensemble activity from the input and output regions of hippocampus while the patients performed a computerized “working memory” task. The data was then sent to USC for nonlinear modeling. About a week after the original recording session (before the patient was scheduled to have the electrodes removed), the WFBMC team again tested the patient, but this time, the recordings from the input areas of hippocampus were fed into an online, real-time version of the nonlinear model, and the output of the model was sent to a device capable of delivering a pattern of minute electrical stimulation pulses to the output areas of hippocampus. Each stimulation pulse was limited to 2 milliseconds and was only 150 microamperes of current, much less than DBS stimulation or even the pulses used by Wilder Penfield in his surgical experiments to map brain function. The stimulation was also limited to occur only during the “encoding” phase of memory on about a third of the test trials. Another third of the trials received a randomized pattern of stimulation that was not produced by the model (and tended to look similar to the fixed frequency stimulation utilized by the other teams mentioned above). The final third of the trials received no stimulation so that they could be directly compared to the stimulated trials.
The result of the stimulation test was a thirty-seven percent improvement on working memory trials in which information was required to be held for up to two minutes. Another version of the task tested memory up to seventy-five minutes, and showed that the stimulation improved retention and recall of memory by thirty-five percent. The randomized stimulation trials did not show any improvement, and even showed a decrease in memory function in some subjects, consistent with the direct hippocampal stimulation tested by UPenn and UCLA. We were also quite encouraged by the fact that the system worked to improve memory even in patients who were already experiencing problems with short-term memory. Given that the nonlinear model was constructed from recordings in the same patient, critics had warned us that we would require perfect memory to train the model, and would not be able to produce positive effects in someone without perfect memory. On the contrary, by identifying the neural “codes” when memory was correct or incorrect, and building the model based on those correct codes, we were able to build-in a “tolerance” to errors that allowed us to improve memory for all of the patients tested.
Both sets of experiments provided results that suggest that it is possible to design and build a brain-interface prosthetic device to improve memory in humans. However, what is really needed to turn these results into a device, and which device is more appropriate for treating the type of memory loss seen in Alzheimer's Disease and other brain injuries and diseases? The answer is that both approaches would produce a valuable memory prosthetic: a device based on the UPenn findings would detect when memory is not functioning properly, and produce a state associated with correct function; whereas a device based on the WFBMC/USC design would be able to by-pass damaged areas of the brain involved in processing memory. However, the UPenn device, like pharmaceutical enhancement, biofeedback, TMS and tDCS, relies on the normal cellular mechanisms of memory being intact. None of these approaches would necessarily help patients who begin to lose neurons and neural circuits due to epilepsy, Parkinson's and advanced stages of Alzheimer's. On the other hand, the WFBMC/USC device concentrates on hippocampus, and may not be applicable to memory functions dependent on brain areas such as prefrontal cortex, or long-term memory beyond the seventy-five minutes tested so far. Ultimately, the designs will evolve as further research is performed, but the current results are exciting and promising as the next step to being able to counter memory loss.

Fixing broken brains requires knowledge of what makes the brain work in the first place. © 2018 Lightspring, image licensed from Shutterstock
Where Do We Go from Here?
As a scientist, what are the next steps? What will turn either proof of concept into a viable prosthetic? And, as an author who dabbles in the science fiction realm, what are the more futuristic implications of this work?
For the research and development, we need more patient testing. We need to change up the memory tasks we use and see whether the principles apply to all types of memory, or just some. We also need to determine how best to convert the algorithms and models into ones that can operate independently for years without external supervision—in other words to create something analogous to a pacemaker which senses, responds, and acts automatically without a human having to make decisions. There is also additional technology needed, such as improved electrodes that fit more electrical sensing into smaller designs, and can remain implanted for years without losing the ability to sense the electrical currents underlying brain activity. We need to determine whether our designs are robust enough to provide solutions for multiple injury or disease states. Finally, we need to always strive for new technologies that allow us a more precise “view” of the function of the brain, especially when we can do it from the outside and not require implanted electrodes and sensors!
From a more speculative approach, what are the implications of this research? Are we truly unlocking all of the abilities of the brain?
First and foremost, you will notice that this essay does not talk about “enhancement” of memory, particularly in subjects with normal memory and the absence of disease or injury. The primary reason is that enhancement was not our goal; our goal was restoration of lost function. The most basic premise of the WFBMC-USC research has been to find a way to bypass a damaged brain area to allow restoration of normal function. We do that by mathematically modeling the function of hippocampus, recording the signals that are naturally input to hippocampus, route that to our model, and then send the output of the model back as a corrected and strengthened information stream using the subject's own codes and signals for memory. This last point is extremely important, because it also means that we are working only with what is already present in the brain under normal, undamaged conditions!
There are, however, some interesting implications for the science fiction fan and futurist: To date, neural-interface prosthetics have come in two types: input to brain, and output to brain. Input prosthetics substitute an artificial signal for the normal sensory signals such as vision and hearing. Thus, retinal implants and cochlear implants are input prosthetics, and as our neuroscience techniques improve and the devices miniaturize, we may even see fully implantable devices such as “bionic eyes” and “bionic ears” popular in TV, movies and books (e.g. Martin Caidin's Cyborg, the basis for TV's The Six Million Dollar Man). On the other hand, output prosthetics turn brain signals into external movement: robotic arms and neurally-controlled limbs are output prosthetics and provide an important link between thoughts and actions, particularly for people who have lost the ability to interact with their environment.
Combining the two types of prosthetic—input and output—how far are we from an artificial body, or even the ability to strap on a mecha (as in Travis Taylor's Tau Ceti Agenda series) or even a spaceship (as in Anne McCaffrey's The Ship Who Sang)? We are starting to see the first signs with Johnny Metheny, a man who lives with a permanently mounted artificial arm that he controls much the same way he would a natural arm [https://www.army.mil/article/167735/darpas_mind_controlled_robotic_arm_does_everything]. As a society we have clear evidence of the need for functional artificial limbs for soldiers, accident victims and cancer survivors who have lost limbs, but what about a whole body? The late Stephen Hawking provided striking evidence that even as his body withered, his mind remained sharp and vital. Again, the advances in brain computer interfaces used for entertainment [https://spectrum.ieee.org/the-human-os/biomedical/bionics/brainy-startup-neurable-unveils-the-worlds-first-braincontrolled-vr-game] show us that the ability to make a difference exists today, and only needs to be put into practice!
There is, however, a third class of prosthetic that is still very early in development, and that is characterized by devices that do not involve the input or output functions of the brain. A “brain-to-brain” prosthetic, such as DBS or the memory prosthetic reported here, suggests the ability to fix broken brains and overcome injury and disease. Over the past ten years, some epilepsy patients have received the option of an implant that senses when a seizure is about to occur, and sends signals that can prevent or lessen the severity of the seizure. As always, science fiction gives us examples, such as Michael Crichton's The Terminal Man, but also cautions scientists and clinicians alike to not be so arrogant as to think we know everything there is to know about the brain! Still, I think that over the next five-to-ten years, we will see an explosion of possibilities for brain-to-brain prosthetics, and will see adoption of those technologies within not much more than that time span.
Brain-to-computer interfaces (BCIs) are simply a specialized case of brain-to-brain prosthetics, where the output “brain” is a synthetic one. The “Neurable” brain-controlled game headset mentioned above is a BCI that has its origins in the use of EEG signals to provide computer control to “locked-in” patients who have lost all voluntary muscle control. By recording the EEG signals associated with viewing images on a computer screen, such devices can be used to track a person's attention, and even allow them to select and "point" at images and letters on a screen to type messages. This is an early phase compared to the full immersion computer interfaces proposed by James A. Hogan in The Genesis Machine and Realtime Interrupt, or the “cyberpunk” genre popularized by William Gibson, Bruce Sterling and others; still, it is a start, and it is a technology that exists now.
The possibilities truly are endless. From the perspective of the current research, I cannot truly imagine which of the present approaches to restoring brain function will be adopted, and which will be discarded or sent back “to the drawing board” for more study. I do know that our memories are at the heart of what it means to be human, and are threatened by head injury, stroke, and diseases that damage the brain. We showed that we could tap into a patient’s own memory content, strengthen it and feed it back to the patient to assist patient's own brain in making correct memory. The ability to preserve and restore memory is one of the most important research directions that I can imagine, and it is this that keeps me coming to work each day.
[Disclaimer: The studies described above have been developed in part with funding from the Defense Advanced Research Projects Agency. The views, opinions and/or findings expressed are those of the author(s) and should not be interpreted as representing the official views or policies of the Department of Defense or the U.S. Government. Approved for public release, distribution unlimited]
Copyright © 2023 by Robert E. Hampson
Dr. Robert Hampson is a neuroscientist with a keen interest in neural prosthetics and human health in space. He is also known to Baen readers and SF convention audiences by his penname "Tedd Roberts," with website here. Dr. Hampson and fellow Baen author and space scientist Les Johnson are the editors of the upcoming Baen anthology Homo Stellaris, a look at the changes both physical and technological that will be wrought on human beings as we head for the stars.